Class 12 Chemistry - Chapter General Principles and Processes of Isolation of Elements NCERT Solutions | State the role of silica in the metallur
State the role of silica in the metallurgy of copper.
During the roasting of pyrite ore, a mixture of FeO and Cu2O is obtained.

The role of silica in the metallurgy of copper is to remove the iron oxide obtained during the process of roasting as 'slag'. If the sulphide ore of copper contains iron, then silica (SiO2) is added as flux before roasting. Then, FeO combines with silica to form iron silicate, FeSiO3 (slag).

More Questions From Class 12 Chemistry - Chapter General Principles and Processes of Isolation of Elements
- Q:-
What is the significance of leaching in the extraction of aluminium?
- Q:-
Write down the reactions taking place in different zones in the blast furnace during the extraction of iron.
- Q:-
Copper can be extracted by hydrometallurgy but not zinc. Explain.
- Q:-
Outline the principles of refining of metals by the following methods:
(i) Zone refining
(ii) Electrolytic refining
(iii) Vapour phase refining
- Q:-
Name the common elements present in the anode mud in electrolytic refining of copper. Why are they so present ?
- Q:-
The reaction,
Cr2O3 + 2Al → Al2O3 + 2Cr (ΔGo = -421kJ)
is thermodynamically feasible as is apparent from the Gibbs energy value. Why does it not take place at room temperature?
- Q:-
How can you separate alumina from silica in bauxite ore associated with silica? Give equations, if any.
- Q:-
Why is the extraction of copper from pyrites more difficult than that from its oxide ore through reduction?
- Q:-
Write chemical reactions taking place in the extraction of zinc from zinc blende.
- Q:-
Describe a method for refining nickel.
Popular Questions of Class 12 Chemistry
- Q:-
For the reaction R → P, the concentration of a reactant changes from 0.03 M to 0.02 M in 25 minutes. Calculate the average rate of reaction using units of time both in minutes and seconds.
- Q:-
Write the formulas for the following coordination compounds:
(i) Tetraamminediaquacobalt (III) chloride
(ii) Potassium tetracyanonickelate(II)
(iii) Tris(ethane-1,2-diamine) chromium(III) chloride
(iv) Amminebromidochloridonitrito-N-platinate(II)
(v) Dichloridobis(ethane-1,2-diamine)platinum(IV) nitrate
(vi) Iron(III) hexacyanoferrate(II)
- Q:-
(i) Write structures of different isomeric amines corresponding to the molecular formula, C4H11N
(ii) Write IUPAC names of all the isomers.
(iii) What type of isomerism is exhibited by different pairs of amines?
- Q:-
Why are solids rigid?
- Q:-
Write any two characteristics of Chemisorption.
- Q:-
Write the structures of the following compounds.
(i) α-Methoxypropionaldehyde
(ii) 3-Hydroxybutanal
(iii) 2-Hydroxycyclopentane carbaldehyde
(iv) 4-Oxopentanal
(v) Di-sec-butyl ketone
(vi) 4-Fluoroacetophenone
- Q:-
Why are pentahalides more covalent than trihalides?
- Q:-
Silver atom has completely filled d orbitals (4d10) in its ground state. How can you say that it is a transition element?
- Q:-
Glucose or sucrose are soluble in water but cyclohexane or benzene (simple six membered ring compounds) are insoluble in water. Explain.
- Q:-
Write structures of the following compounds:
(i) 2-Chloro-3-methylpentane
(ii) 1-Chloro-4-ethylcyclohexane
(iii) 4-tert. Butyl-3-iodoheptane
(iv) 1,4-Dibromobut-2-ene
(v) 1-Bromo-4-sec. butyl-2-methylbenzene
Recently Viewed Questions of Class 12 Chemistry
- Q:-
Why do soaps not work in hard water?
- Q:-
Arrange the following compounds in increasing order of their boiling points.
CH3CHO, CH3CH2OH, CH3OCH3, CH3CH2CH3
- Q:-
Name the following compounds according to IUPAC system of nomenclature:
(i) CH3CH(CH3)CH2CH2CHO
(ii) CH3CH2COCH(C2H5)CH2CH2Cl
(iii) CH3CH=CHCHO
(iv) CH3COCH2COCH3
(v) CH3CH(CH3)CH2C(CH3)2COCH3
(vi) (CH3)3CCH2COOH
(vii) OHCC6H4CHO-p
- Q:-
Write structural formulas and names of four possible aldol condensation products from propanal and butanal. In each case, indicate which aldehyde acts as nucleophile and which as electrophile.
- Q:-
Complete each synthesis by giving missing starting material, reagent or products
(i)
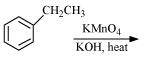
(ii)

(iii)

(iv)

(v)

(vi)
- Q:-
Although phenoxide ion has more number of resonating structures than carboxylate ion, carboxylic acid is a stronger acid than phenol. Why?
- Q:-
Calculate the osmotic pressure in pascals exerted by a solution prepared by dissolving 1.0 g of polymer of molar mass 185,000 in 450 mL of water at 37°C.
- Q:-
Write the structures of products of the following reactions;
(i)

(ii)

(iii)

(iv)

- Q:-
Predict the products of the following reactions:
(i)

(ii)

(iii)

(iv)

- Q:-
Give the IUPAC names of the following compounds:
(i) PhCH2CH2COOH (ii) (CH3)2C=CHCOOH
(iii)
 (iv)
(iv) 
1 Comment(s) on this Question
If FeO adds with FeSiO3, what will happen?!?
- All Chapters Of Class 12 Chemistry
- All Subjects Of Class 12
